Sodium Methyl Cocoyl Taurate in Toothpaste: Benefits & Safety
- David Chen, DDS
- Dec 22, 2023
- 4 min read
The purpose of Sodium Methyl Cocoyl Taurate (SMCT) in toothpaste is to help make it easier to clean your teeth because it's a surfactant. If you don't know what a surfactant is, another description for this ingredient would be a soap/detergent.
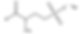
In regards to its safety, it's on the safer side especially when compared to other surfactants such as SLS. It's considered as a less harsh alternative to it.
Below is a concise summary with things to know about it.
Sodium Methyl Cocoyl Taurate in toothpaste overview:
Description: Coconut fatty acid derived cleansing agent.
Benefits: Foaming; Wetting; Removes food/plaque/stains.
Side effects: Dermal irritation.
Safety: Safe for cosmetic use; No oral toxicity; No carcinogenicity.
Description
SMCT is a coconut fatty acid derived anionic surfactant with excellent lathering and cleaning properties. That means it is a natural ingredient and its use is as a soap to clean with.
Properties:
Soft off-white appearance
Mild fatty smell.
Common uses:
Shampoos/Conditioners
Liquid soaps
Face lotions
Skin creams
Bubble baths
Toothpaste
Detergents
It is a very common ingredient in personal hygiene and cosmetic products.
Benefits/Functions in toothpaste
Sodium methyl cocoyl taurate is a surfactant and what it does in a toothpaste is similar to a soap/detergent. The gist of it is that it helps to clean your teeth.

Effects in toothpaste:
Cleansing. As with all soaps, it assists in the removal of food, plaque, bacteria, and stains from the tooth surfaces. It helps loosens all of them from the teeth and that makes them easier to remove.
Wetting. It helps wet the surfaces of the teeth by spreading the toothpaste across them and reducing surface friction. It facilitates the cleaning process.
Foaming. It creates foam to help the toothpaste spread and reach into every nook and cranny of the teeth. It also reduces the amount of toothpaste needed for each brushing session.
Consequences of removing SMCT and all other surfactants from toothpaste:
Reduced cleaning efficacy. If you remove the detergent, it will not clean the teeth as well. It is similar to cleaning dinner plates with no soap.
No foaming. There will be no-to-minimal foaming. This effect is readily observable in SLS-free toothpastes.
Discomfort while brushing. SMCT helps to wet the surfaces of the teeth so that the toothbrush glides easily. Without it, it would be akin to dry brushing with can be uncomfortable.
Use a lot more toothpaste. Due to less foaming, you will need to use a lot more toothpaste to get the same clean sensation.
Vs SLS
SMCT is considered more natural and less harsh than SLS in toothpaste but there are also a couple of other differences.
Differences:
More natural. The cocoyl in SMCT means it is derived from coconut, a whole food. In other words, it is a more natural ingredient.
Less harsh. SMCT doesn't strip as much of the natural mouth oils.
Less foaming. The most foaming toothpastes all use SLS.
Reduce cleaning efficacy. SMCT still cleans but not as effectively as SLS. Studies have shown that the bacterial survival rate on toothbrushes were significantly lower for SLS vs SMCT.
Below are various images showing various toothpastes with different surfactants:
Image 1 - Amount of foaming with no SLS and no SMCT.
Image 2 - Amount of foaming with SMCT.
Image 3 - Amount of foaming with SLS.
Objectively... the stronger the surfactant the more foaming it creates.
Side note: I find it fascinating that surfactants in toothpaste can kill bacteria. Although it seems like the harsher sodium lauryl sulfate IS more effective at it.
Vs Cocamidopropyl Betaine
Cocamidopropyl betaine is another surfactant that is used as an alternative to SLS. What distinguishes cocamidopropyl betaine from sodium methyl cocoyl taurate is its bitter taste and greater foaming ability.
It works very well but it doesn't taste the very pleasant. For that reason, it is strategically mixed with SMCT to help reduce the poor taste.
Adverse effects
In regards to use in toothpaste, the only adverse effect for sodium methyl cocoyl taurate is dermal irritation. Everything else was trivial and of no concern.
However, these studies were performed specifically on human skin and not inside the mouth. I did not find any research that showed soft tissue irritation within the oral cavity.
Nonetheless, SMCT is a coconut derived ingredient and while it is mostly safe, it can occasionally cause very mild skin irritation. In my opinion, there is no reason why that wouldn't apply to the mouth as well.
Safety
Sodium methyl cocoyl taurate is relatively safe to use in toothpaste and also in cosmetic products in general. That was the conclusion the Cosmetic Ingredient Review came to, it was safe for cosmetic use.
Research findings:
No oral toxicity. No adverse effects were observed via oral administration of it to mice for 28 days.
No genotoxicity. It is not genotoxic in mammalian chromosomes.
No carcinogenicity. No published and unpublished studies were found.
Dermal irritation. Out of 11 test subjects, 2 had slight to definite reddening of the skin. The other 9 had no reactions at all.
Overall, the conclusion that I came to was that this ingredient is safe to use. The only potential side effect was the skin irritation but you know it isn't uncommon for people to have reactions to coconut oil.
If your body can't tolerate it, that is a personal/individual problem. It just means that this ingredient isn't the right one for you personally.
Verdict
In my opinion, sodium methyl cocoyl taurate is safe to use in toothpaste and I do consider it as a viable alternative to SLS. I would consider using it in my own personal toothpaste formulation.
It is safe: This ingredient is derived from coconuts
Viable alternative: It is a more natural alternative to using sodium lauryl sulfate which can be too drying/harsh on the oral tissues. After all it is derived from coconuts which is a proven natural moisturizer.
You need to have some type of surfactant in a toothpaste because it helps it lather and it makes for a more pleasant brushing experience. Without it, you'd be dry brushing your teeth which can be painful.
That is why this ingredient is needed and as of the moment it does appear to be one of the better detergents on the market. With that, this toothpaste ingredient has my seal of approval along with afterva's.






